Musiyano wakakosha pakati peNitrate uye Nitrite ndeyekuita maatomu matatu eokisijeni akasungwa kuenda kuNitrogen Atom asi nitrite ine maatomu maviri eokisijeni akasungwa kune nitrogen atomu.
Ose ari maviri nitrate uye nitrite ari maloranic ansign anosanganisira nitrogen uye oksijeni maatomu. Zvese izvi zvioni zvine chekupomerwa kwemagetsi. Zvinonyanya kuitika seanonwa kwemunyu. Kune mamwe mutsauko pakati pekutungamira naNitrite; Isu tichave tichikurukura izvo misiyano iri muchinyorwa chino.
Chii chinonzi nitrate?
Nitrate isorganic anion ine makemikari formula no3-. Icho chinonzwikwa chollatomic icho chine maatomu mana; imwe nitrogen atom uye matatu oksijeni maatomu. Anon ane -1 pesell. Iyo Malar Huru yeiyi IONS ndeye 62 G / MOL. Zvakare, ichi chionana chinotorwa kubva kune yayo yemanzwi acid; nitric acid kana hno3. Mune mamwe mazwi, nitrate ndiyo conjugate chigadziko che nitric acid.
Muchidimbu, nitrate ion ine imwe nitrogen atomu pakati peiyo inosunga maatomu matatu eokisijeni kuburikidza necavent cemical bhaved. Paunenge uchifunga nezvekugadzira makemikari echioni ichi, ine zvirevo zvitatu zvisina kufanana Nekudaro, geometry yemamorekuru ndeye trigonal planar. Imwe neimwe oxygen atom inotakura a - 2/3 muridzi, iyo inopa mari yakawanda yeanon se -1.
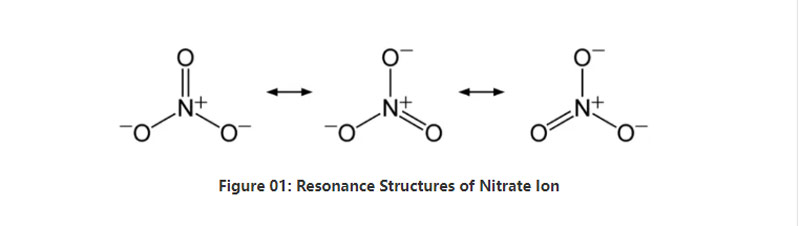
Panguva yekumanikidza uye tembiricha, dzinenge dzese munyu dzemunyu dzine nyuchi idzi dzinopera mumvura. Tinogona kuwana zvoga zvinoitika zvichireva runyararo pasi sezvinoita deposits; nitrative deposits. Iyo inonyanya kuve nesodium nitrate. Uyezve, hutachiona hwehutachiona hunogona kuburitsa nitrate ion. Imwe yemashandisirwo makuru eiyo nitrate salts iri mukugadzirwa kwefetiraiza. Uyezve, zvinobatsira semuenzaniso oxdizizing mumatanho ekuputika.
Chii chinonzi nitrite?
Nitrite inzvimbo yemukati ine munyu ine makemari formula no2-. Izvi zvinonyadzisa chifananidzo chinonzwika, uye ine imwe nitrogen atomu yakasungwa kumaatomu maviri eokisijeni aine maviri akafanana asina kuunganidzwa makereke emakemikari. Nekudaro, iyo nitrogen atomu iri pakati pemamorekuru. Anon ane -1 pesell.
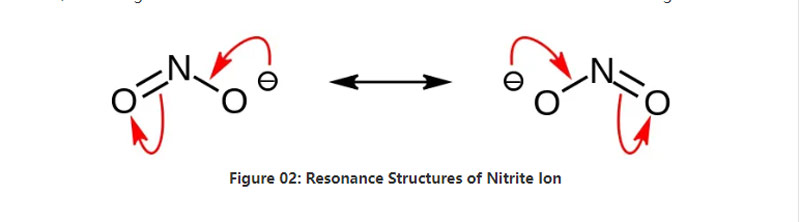
Iyo Malar Huru yeAnion is 46.01 G / Mol. Zvakare, ichi chinonzwika chinobva kune iyo nitrow acid kana hno2. Nekudaro, iyo iyo conjugate chigadziko cheiyo nitrow asidhi. Naizvozvo, isu tinogona kuburitsa nitrite salts inyatso vashama kuburikidza nekupfuura nitrous nhumbu mumvura inonzi Sodium Hydroxide Solution. Uyezve, izvi zvinoburitsa sodium nitrite izvo zvatinogona kunatsa kuburikidza neRetrystallization. Uyezve, nitrite munyu sodium nitrite inobatsira pakuchengetedza chikafu nekuti inogona kudzivirira chikafu kubva mukukura kwemicrobial.
Ndeupi musiyano uripo pakati peNitrate naNitrite?
Nitrate isorganic anion ane makemikari formula no3- nepo nitrite inzvimbo inormanic ine yemakemikari emafomu kwete2-. Naizvozvo, musiyano wekutanga pakati peNitrate uye nitrite iri pamusoro pekuumbwa kwemakemikari ezvipenga zviviri. Ndiko kuti; Musiyano wakakosha pakati peNitrate uye Nitrite ndeyekuita maatomu matatu eokisijeni akasungwa kuenda kuNitrogen Atom asi nitrite ine maatomu maviri eokisijeni akasungwa kune nitrogen atomu. Uyezve, nitrate ion inotorwa kubva kune yayo conjugate acid; iyo nitric acid, nepo nitrite ion ichitorwa kubva kune nitrous acid. Semumwe musiyano wakakosha pakati peNitrate uye nitrite ions, tinogona kutaura kuti nitrate ndeye oxdizing mumiriri nekuti inogona kuderedzwa chete nepo nitrite inogona kuita seya ollidizing uye kuderedza mumiririri.
Kutumira Nguva: May-16-2022





